Abstract
Peptide therapy has become a standard offering in functional and longevity medicine practices. It has also become a standard source of clinical liability, regulatory scrutiny, and patient safety incidents. The reason is structural: peptides involve complex dosing, patient-specific contraindications, drug-peptide interactions, and a knowledge base that evolves faster than individual clinicians can track.
Automated risk flagging is not a luxury feature in peptide prescribing infrastructure — it is a safety necessity. This brief provides an operational implementation guide for building or deploying automated risk detection systems in clinical practice. We specify what risks to flag, how to flag them without creating alert fatigue, what data infrastructure is required, and how to integrate risk flags into clinical workflow without adding cognitive burden to the prescribing physician.
This is a practical, step-by-step implementation guide designed for medical directors, clinical operators, and compliance officers in functional medicine practices currently prescribing peptides or planning to do so.
I. The Case for Automated Risk Flagging: Why Manual Review Is Insufficient
We begin with a question that some clinicians will find uncomfortable: if you are prescribing peptides to 50, 100, or 200 patients, can you reliably remember — at the moment of prescription — every patient’s current medication list, every relevant lab abnormality, every documented allergy, and every peptide-drug interaction that might produce harm?
The honest answer is no. Human working memory is not designed for this task. The average physician can hold 5-7 discrete pieces of information in active working memory at any given time [1]. A comprehensive contraindication check for a single peptide prescription might require reviewing 15-20 data points. This is not a cognitive failure — it is a cognitive impossibility.
The question is not 'Are my clinicians competent?' The question is 'Is the system designed to make competence systematically achievable at scale?' If the answer is no, you have a system failure, not a people failure.
Automated risk flagging solves a specific problem: it externalizes the contraindication checking and pattern recognition functions that cannot reliably occur in human working memory, and surfaces those risks at the exact moment when clinical decision-making is occurring. Done correctly, it does not replace clinical judgment. It informs it.
1.1 What Automated Risk Flagging Is Not
Before we specify what to build, we must clarify what not to build. The following are common failure modes in clinical alert systems:
Alert Fatigue Generators (Anti-Patterns to Avoid) Pop-up alerts that interrupt workflow for low-severity risks — these train clinicians to ignore all alerts. Alerts that trigger on outdated data — flagging a medication the patient stopped taking six months ago destroys trust in the system. Non-specific warnings — 'Use caution with this peptide' is not actionable guidance. Alerts without suppression options — if a clinician has reviewed and acknowledged a risk, the system should not re-alert on every subsequent interaction. Alerts that do not distinguish severity — a true contraindication and a mild caution should not have identical visual presentation.
The objective is surgical precision: flag only risks that are clinically actionable, present them in proportion to their severity, and make acknowledgment or override a documented, intentional decision rather than a reflexive click-through.
II. The Risk Taxonomy: What to Flag and at What Severity
Not all risks are equivalent. A documented peptide allergy is categorically different from a mild drug interaction that might require dose adjustment. The risk flagging system must encode this distinction at the architectural level.
What follows is the comprehensive risk taxonomy organized by category and severity.
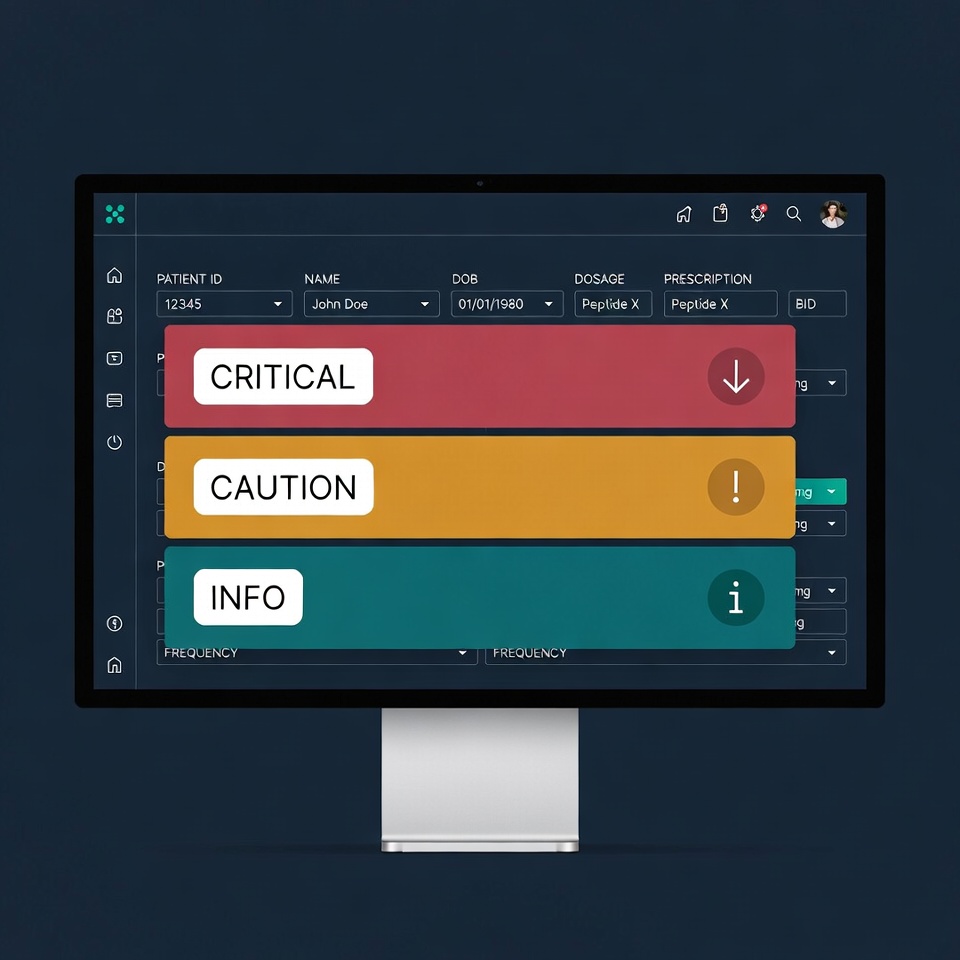
2.1 RED Alerts (Critical / Prescription-Blocking)
Category 1: Documented Allergies and Prior Adverse Reactions
- Trigger Condition: Patient has documented allergy to the specific peptide being prescribed, OR patient has documented severe adverse reaction to a peptide in the same structural class.
- Alert Text: ‘CRITICAL: Patient has documented allergy to [peptide name]. Prescription blocked. Override requires explicit justification and attending physician approval.’
- Data Requirement: Allergy list must be current (reviewed within past 30 days) and include peptide-specific entries, not just traditional drug allergies.
Category 2: Pregnancy and Lactation
- Trigger Condition: Patient is documented as pregnant OR currently breastfeeding AND peptide is classified as Pregnancy Category C, D, or X (or lacks pregnancy safety data).
- Alert Text: ‘CRITICAL: Patient is pregnant/breastfeeding. [Peptide name] has insufficient pregnancy safety data. Prescription blocked unless explicit maternal-fetal medicine consultation documented.’
- Data Requirement: Pregnancy status must be current (updated within 60 days for women of childbearing age). Peptide pregnancy classification must be maintained in formulary database.
Category 3: Severe Organ Dysfunction
- Trigger Condition: Patient has eGFR <30 (Stage 4-5 CKD) OR ALT/AST >5x ULN (severe hepatic dysfunction) AND peptide requires renal or hepatic clearance.
- Alert Text: ‘CRITICAL: Patient has severe renal/hepatic dysfunction. [Peptide name] clearance severely impaired. Prescription blocked unless nephrology/hepatology clearance documented.’
- Data Requirement: Renal and liver function labs must be current (<90 days). Peptide clearance pathway must be documented in formulary.
2.2 YELLOW Alerts (Caution / Requires Acknowledgment)
Category 4: Moderate Organ Dysfunction
- Trigger Condition: eGFR 30-59 (Stage 3 CKD) OR ALT/AST 2-5x ULN (moderate hepatic impairment) AND peptide requires renal/hepatic clearance.
- Alert Text: ‘CAUTION: Patient has moderate renal/hepatic impairment. Consider 25-50% dose reduction for [peptide name]. Enhanced monitoring recommended.’
- Suggested Action: System suggests modified dosing range. Clinician must acknowledge or override.
Category 5: Drug-Peptide Interactions
- Trigger Condition: Patient is on medication known to interact with prescribed peptide (examples: anticoagulants + BPC-157, immunosuppressants + Thymosin Alpha-1).
- Alert Text: ‘CAUTION: Patient is on [drug name] which may interact with [peptide name]. Interaction type: [mechanism]. Monitoring recommendation: [specific labs/vitals].’
- Data Requirement: Current medication list (reviewed within 7 days). Drug-peptide interaction database maintained and version-controlled.
Category 6: Polypharmacy and Age-Related Risk
- Trigger Condition: Patient is on ≥5 concurrent medications AND age ≥65 OR age ≤25.
- Alert Text: ‘CAUTION: Patient has polypharmacy and is in higher-risk age group. Consider starting at 50% standard dose with slower titration.’
- Rationale: Both advanced age and polypharmacy independently increase adverse event risk. Combined, they warrant conservative dosing.
Category 7: Peptide Stacking (Combination Protocols)
- Trigger Condition: Patient is currently prescribed ≥2 peptides simultaneously AND new peptide being added.
- Alert Text: ‘CAUTION: Patient is currently on [list existing peptides]. Adding [new peptide] creates a multi-peptide protocol. Enhanced monitoring recommended. Review interaction matrix.’
- Data Requirement: Active peptide prescription tracking. Peptide-peptide interaction matrix (even if limited data — flag unknown interactions as cautions).
2.3 BLUE Alerts (Informational / Optional Acknowledgment)
Category 8: Lab Value Trends
- Trigger Condition: Relevant biomarker (creatinine, liver enzymes, inflammatory markers) is trending upward over past 2-3 measurements, even if still within normal range.
- Alert Text: ‘INFO: Patient’s [biomarker] has increased from [value 1] to [value 2] over past [timeframe]. Consider monitoring more closely if prescribing [peptide].’
- Value: Surfacing trends prevents waiting until a value is frankly abnormal to notice deterioration.
Category 9: Prior Peptide Exposure
- Trigger Condition: Patient has previously been prescribed this peptide with documented outcome (positive or negative).
- Alert Text: ‘INFO: Patient previously used [peptide] from [start date] to [end date]. Documented outcome: [response/tolerance/adverse event]. Review prior notes.’
- Value: Institutional memory — prevents re-prescribing a peptide that the patient did not tolerate or informing dosing based on prior response.
III. The Data Infrastructure Requirements
3.1 Required Data Fields and Freshness Standards
The following data fields must be captured, structured, and maintained to current standards:
If any required data field is missing or stale, the system should flag the data gap to the clinician BEFORE generating peptide recommendations or risk alerts. This is a ‘fail-safe’ design: the system does not pretend to provide safety checking when it lacks the data to do so.
3.2 The Drug-Peptide Interaction Database
This is the most operationally complex data requirement. Unlike drug-drug interactions — which are cataloged in mature databases like Lexicomp and Micromedex — peptide-drug and peptide-peptide interactions are poorly documented, frequently speculative, and evolving as new data emerges.
HolistiCare maintains a curated peptide interaction library that is updated as new evidence emerges. However, practices implementing their own risk flagging systems should expect to build and maintain this database internally. Here is the minimum viable structure:
Even a partially complete interaction database is better than none. Start with the highest-volume peptides in your practice and the highest-risk drug classes (anticoagulants, immunosuppressants, antihypertensives). Expand iteratively.
3.3 Integration with Existing EHR Infrastructure
Most functional medicine practices operate on legacy EHRs (Cerbo, Practice Better, Charm) that were not designed for peptide-specific clinical decision support. HolistiCare’s Clinical Intelligence Layer is purpose-built to sit atop these systems and provide the structured data layer that enables automated risk flagging.
For practices not using HolistiCare, integration can be achieved through:
- HL7/FHIR data feeds: If your EHR supports it, configure automated data extracts for allergies, medications, labs, and active prescriptions.
- Manual structured data entry: At minimum, maintain a parallel peptide-specific patient registry in a structured database (even a well-designed spreadsheet is better than chart notes).
- Middleware solutions: API-based integration tools (Zapier, custom scripts) can pull data from EHR APIs and feed it into a risk-flagging engine.
The critical principle: automated risk flagging requires structured data. If your patient data lives in unstructured chart notes, risk flagging cannot occur. This is not a limitation of AI or software — it is a limitation of data architecture.
Related Resource: For detailed technical specifications on HolistiCare’s peptide platform architecture, see: HolistiCare Peptide Therapy Platform
IV. Workflow Integration: Where and How to Surface Risk Flags
The most sophisticated risk detection system is clinically useless if it interrupts workflow, creates cognitive burden, or trains clinicians to ignore alerts. This section specifies how to integrate risk flags into clinical workflow in a way that enhances rather than obstructs decision-making.
4.1 The Three-Checkpoint Model
We recommend surfacing risk flags at three distinct checkpoints in the peptide prescribing workflow:
- Checkpoint 1: Pre-Prescription Review (Passive Display). When the clinician opens a patient chart to consider peptide therapy, the system displays a risk summary panel showing: current medications, recent labs, active peptide prescriptions, and any standing contraindications. This is non-interruptive — it is information available for review if the clinician chooses to consult it.
- Checkpoint 2: Active Prescription Entry (Contextual Alert). When the clinician selects a specific peptide and dosing regimen, the system runs the full risk taxonomy and surfaces any triggered alerts (RED/YELLOW/BLUE) in a modal dialog that must be acknowledged before proceeding. This is the primary safety checkpoint.
- Checkpoint 3: Post-Prescription Monitoring (Scheduled Re-Check). After prescription, the system schedules follow-up risk re-checks at defined intervals (e.g., 2 weeks, 1 month, 3 months) and flags any new lab abnormalities or medication changes that alter the risk profile.
This three-checkpoint model balances proactive risk awareness (Checkpoint 1), mandatory safety verification (Checkpoint 2), and ongoing surveillance (Checkpoint 3) without overwhelming the clinician with alerts at every interaction.
4.2 Alert Presentation Design Principles
How an alert is presented matters as much as what it says. Poor presentation creates alert fatigue. Good presentation enables rapid, informed decision-making.
Best Practices for Alert UI/UX (Use color coding consistently: RED = stop, YELLOW = caution, BLUE = info. Do not deviate. Lead with the risk, not the explanation. 'CRITICAL: Severe renal dysfunction detected' not 'The system has identified a potential contraindication related to...' Provide one-click access to supporting data. Alert says 'eGFR 28'. Clicking it shows the full lab trend over 6 months. Offer specific clinical guidance, not vague warnings. 'Reduce dose by 50% and recheck creatinine in 2 weeks' not 'Use caution.' Allow alert suppression with justification. If a clinician overrides a YELLOW alert, require them to document why (free text, 1-2 sentences). This creates an audit trail and encourages deliberate decision-making. Do not re-alert on acknowledged risks unless new data changes the risk profile. Once a clinician has reviewed and accepted a drug interaction, the system should not re-surface that alert on every subsequent visit unless a new medication is added.
4.3 Mobile and Point-of-Care Considerations
Many functional medicine clinicians conduct patient consultations via telemedicine or review charts on mobile devices. Risk flagging systems must be responsive and functional on small screens without compromising information density.
Key mobile design requirements:
- Alerts must be readable and actionable on a 5-inch screen
- Supporting data (labs, medication lists) should be accessible via expandable panels, not requiring navigation away from the alert
- Voice-to-text justification entry for alert overrides (typing multi-sentence justifications on mobile is prohibitive)
- Offline functionality: if connectivity is lost mid-consultation, the system should cache recent patient data and allow prescribing with a ‘sync pending’ flag rather than blocking workflow
IV. The Commercial and Ethical Argument for Restrictive Governance
We anticipate the objection that this governance architecture is too restrictive — that it slows down clinical workflow, reduces the apparent value of the AI, and makes the tool less competitive against faster, more permissive alternatives.
This objection is correct on the first two counts and catastrophically wrong on the third.
4.1 Why Speed Is Not the Correct Optimization Target
An AI tool that generates a peptide recommendation in 2 seconds is not more valuable than one that generates the same recommendation in 10 seconds if the 2-second version omits contraindication checking, evidence grading, or decision logging. Speed is a user experience optimization. Safety is a clinical obligation. They are not equivalent priorities.
The clinician who wants fast recommendations without governance can use Google and a dosing calculator. The clinician who wants decision support that survives regulatory scrutiny, reduces liability exposure, and genuinely improves clinical outcomes should expect — and require — the governance overhead we have described.
4.2 The Regulatory Arbitrage Play
We are building for a market that does not yet exist but will. The current peptide therapy market operates with minimal regulatory oversight. That will not last. The FDA has signaled increased scrutiny of compounded peptides. State medical boards are beginning enforcement actions against physicians making unsupported therapeutic claims. Insurance carriers are tightening coverage criteria for off-label prescribing.
When enforcement increases — and it will — the AI tools that were built for speed rather than safety will become liabilities. The clinicians using those tools will face disproportionate regulatory risk. The tools that were built with restrictive governance from the outset will be the only ones that remain commercially and legally viable.
This is not fearmongering. It is pattern recognition. Every high-growth, under-regulated healthcare market eventually faces a regulatory correction. The operators who anticipated that correction and built accordingly capture the post-correction market. The operators who optimized for the pre-correction environment do not.
4.3 The Ethical Case Is Also the Commercial Case
There is a more fundamental argument. We are physicians. We were trained to a professional standard that places patient welfare above commercial optimization. An AI tool that increases prescription volume at the cost of patient safety is not a clinical innovation — it is a violation of our fiduciary duty to patients.
The market will eventually price integrity correctly. The practice that can demonstrate — with audit logs, evidence trails, and outcome data — that its AI-assisted peptide protocols are safer, more evidence-based, and more carefully monitored than those of competitors will command higher fees, attract higher-quality patients, and survive regulatory enforcement that eliminates less rigorous competitors.
Restrictive governance is not a compromise of commercial viability. It is the structural foundation of sustainable competitive advantage in a market heading toward regulatory maturity.
V. Governance, Audit, and Continuous Improvement
Deploying a risk flagging system is not a one-time implementation. It is an ongoing governance process that requires monitoring, refinement, and accountability.
5.1 The Alert Performance Dashboard
Every risk flagging system should generate a monthly performance report for clinical leadership review. Key metrics:
This dashboard is not a performance evaluation tool for individual clinicians. It is a system performance tool. High override rates indicate that the alert rules need refinement, not that clinicians are non-compliant.
5.2 Quarterly Risk Taxonomy Review
The risk taxonomy described in Section II should be reviewed and updated quarterly. New evidence emerges. New peptides enter the formulary. New drug interactions are documented. The taxonomy must evolve accordingly.
The quarterly review process should include:
- Review of all adverse events in the prior quarter: Were any predictable by existing risk flags? If not, should new flags be added?
- Review of alert override justifications: Are clinicians consistently overriding certain alerts for sound clinical reasons? If so, downgrade severity or remove the alert.
- Literature review: Has new evidence emerged on peptide safety, contraindications, or interactions? Update interaction database and alert logic accordingly.
- Clinician feedback session: Solicit structured feedback from prescribing physicians on alert utility, false positive rate, and workflow integration. Iterate.
5.3 Regulatory and Medicolegal Considerations
Automated risk flagging systems create both protection and exposure from a liability perspective. Here is what you need to document:
Medicolegal Requirements for Risk Flagging Systems Maintain version-controlled documentation of all risk flagging rules, including date implemented and evidence basis. Log all alerts generated and all clinician responses (acknowledged, overridden, justification provided). These logs must be immutable and auditable. Include risk flagging system usage in informed consent language: 'Our clinic uses automated clinical decision support tools to identify potential medication risks. Final prescribing decisions remain with your physician.' Do NOT represent the system as 'AI-powered' or 'intelligent' in patient-facing materials unless you are prepared to defend its performance in litigation. Terms like 'automated safety checks' or 'clinical decision support' are safer. Establish a documented escalation protocol for when alerts are overridden: Does an override require attending physician co-signature? Does it trigger enhanced monitoring protocols? Conduct annual third-party audits of alert accuracy and system performance. External validation is a powerful medicolegal defense.
Related Resource: For comprehensive governance frameworks for AI-assisted peptide prescribing, see our detailed brief: Human-in-the-Loop: AI Governance for Peptide Therapy
VI. Implementation Roadmap: A Phased Deployment Plan
Deploying automated risk flagging is not an all-or-nothing proposition. We recommend a phased implementation that starts narrow and expands as data infrastructure matures and clinician adoption stabilizes.
Phase 1: Foundation (Months 1-2)
Objective: Build the minimum viable data infrastructure and implement RED alert functionality only.
- Audit current data completeness: What % of patients have current allergy lists, medication lists, recent labs? Identify gaps.
- Implement structured data capture for mandatory fields: Allergy list, current medications, pregnancy status, renal function, liver function.
- Build or deploy RED alert rules only: Documented allergies, pregnancy contraindications, severe organ dysfunction.
- Configure alert UI with blocking behavior: RED alerts prevent prescription unless override with justification.
- Train clinical staff on new workflow: Why alerts exist, how to respond, what documentation is required for overrides.
Phase 2: Expansion (Months 3-4)
Objective: Add YELLOW alerts and begin interaction database development.
- Implement YELLOW alert rules: Moderate organ dysfunction, age/polypharmacy cautions, peptide stacking alerts.
- Build initial drug-peptide interaction database: Start with top 10 prescribed peptides and top 20 co-prescribed medications.
- Deploy YELLOW interaction alerts: Test with small cohort of clinicians before full rollout.
- Collect feedback on alert frequency and utility: Adjust sensitivity if false positive rate >10%.
Phase 3: Refinement (Months 5-6)
Objective: Add BLUE informational alerts and implement continuous monitoring.
- Implement BLUE alert rules: Lab trends, prior peptide exposure history.
- Deploy post-prescription monitoring: Scheduled risk re-checks at 2 weeks, 1 month, 3 months.
- Generate first quarterly performance dashboard: Review metrics with clinical leadership.
- Conduct first quarterly risk taxonomy review: Update alert rules based on adverse event data and clinician feedback.
Phase 4: Optimization (Month 7+)
Objective: Continuous improvement, expanded interaction database, and preparation for external audit.
- Expand interaction database coverage to full formulary.
- Implement alert suppression logic for recurring low-value alerts.
- Integrate risk flagging data with outcome tracking: Correlate alert overrides with adverse events.
- Prepare for third-party audit of system accuracy and compliance documentation.
VII. Conclusion: Safety as Infrastructure, Not Aspiration
Patient safety in peptide therapy is not achieved through clinician vigilance alone. Vigilance is a virtue, but it is not a system. Systems fail when they depend on human perfection rather than architectural safeguards.
Automated risk flagging is one such safeguard. It does not replace clinical judgment — it supports it. It does not eliminate liability — it documents the exercise of appropriate clinical diligence. It does not prevent all adverse events — it reduces the frequency of preventable ones.
The practices that deploy risk flagging systems now — before regulatory enforcement mandates it, before a sentinel adverse event forces it — will have the operational infrastructure that others will spend years building under crisis conditions. This is not theoretical risk mitigation. This is operational readiness.
Build it methodically. Build it transparently. Build it with the assumption that every alert, every override, and every clinical decision will eventually be reviewed by someone with subpoena power. If that assumption makes you uncomfortable, your system needs more work.
HolistiCare exists to make that work systematically achievable. The infrastructure described in this brief is not aspirational — it is operational, deployed, and refining itself daily across our partner practices.
Safety is not a feature. It is the foundation.
References
[1] Miller, G. A. (1956). The Magical Number Seven, Plus or Minus Two: Some Limits on Our Capacity for Processing Information. Psychological Review, 63(2), 81-97.
[2] Phansalkar, S., van der Sijs, H., Tucker, A. D., et al. (2013). Drug-drug interactions that should be non-interruptible in order to reduce alert fatigue in electronic health records. Journal of the American Medical Informatics Association, 20(3), 489-493.
[3] Ash, J. S., Sittig, D. F., Campbell, E. M., et al. (2007). Some Unintended Consequences of Clinical Decision Support Systems. AMIA Annual Symposium Proceedings, 2007, 26-30.
[4] Nanji, K. C., Slight, S. P., Seger, D. L., et al. (2014). Overrides of medication-related clinical decision support alerts in outpatients. Journal of the American Medical Informatics Association, 21(3), 487-491.
[5] Kuperman, G. J., Bobb, A., Payne, T. H., et al. (2007). Medication-related Clinical Decision Support in Computerized Provider Order Entry Systems: A Review. Journal of the American Medical Informatics Association, 14(1), 29-40.
[6] van der Sijs, H., Aarts, J., Vulto, A., & Berg, M. (2006). Overriding of drug safety alerts in computerized physician order entry. Journal of the American Medical Informatics Association, 13(2), 138-147.
[7] Bates, D. W., Kuperman, G. J., Wang, S., et al. (2003). Ten Commandments for Effective Clinical Decision Support: Making the Practice of Evidence-based Medicine a Reality. Journal of the American Medical Informatics Association, 10(6), 523-530.
[8] Institute of Medicine. (2000). To Err is Human: Building a Safer Health System. National Academies Press.
[9] Weingart, S. N., Toth, M., Sands, D. Z., et al. (2003). Physicians’ decisions to override computerized drug alerts in primary care. Archives of Internal Medicine, 163(21), 2625-2631.
[10] Sittig, D. F., & Singh, H. (2010). A new sociotechnical model for studying health information technology in complex adaptive healthcare systems. Quality and Safety in Health Care, 19(Suppl 3), i68-i74.
Legal & Medical Disclaimer:
This document is produced for educational and informational purposes by HolistiCare.io and does not constitute medical advice, legal counsel, regulatory guidance, or clinical practice standards. The risk flagging taxonomies, alert severity classifications, and implementation frameworks presented are illustrative models intended for educational discussion among healthcare professionals and clinical system administrators. HolistiCare.io does not guarantee that implementation of the described risk flagging systems will prevent adverse events, eliminate liability exposure, or ensure regulatory compliance in all jurisdictions. Clinical decision support systems must be developed and deployed in accordance with applicable FDA regulations (21 CFR Part 11 for electronic records, Software as a Medical Device guidance), state medical board requirements, HIPAA standards, and institutional policies. All clinical prescribing decisions remain the sole responsibility of the licensed prescribing physician. Automated alerts are decision support tools, not substitutes for clinical judgment. Practices implementing risk flagging systems are advised to consult qualified legal, regulatory, compliance, and clinical risk management professionals. The interaction databases, alert rules, and clinical protocols described must be validated for accuracy and appropriateness within each practice’s specific clinical context. HolistiCare.io is a clinical intelligence software company and does not provide direct clinical services, legal advice, or regulatory consulting.





What do you think?
[…] Related Resource: For automated risk detection and patient safety monitoring infrastructure: Peptide Patient Safety: Automated Risk Flags […]
[…] Related Resource: For automated safety monitoring and compliance documentation: Peptide Patient Safety: Automated Risk Flags […]
Comments are closed.