Abstract
AI-assisted clinical decision support holds enormous promise for peptide therapy — optimizing dosing, detecting contraindications, correlating biomarkers, and improving prescribing safety. It also holds enormous risk if deployed without rigorous governance and explicit human-in-the-loop checkpoints.
This operational guide translates the Human-in-the-Loop (HITL) governance framework into an actual Standard Operating Procedure (SOP) for clinical practice. We specify the end-to-end workflow from patient intake through prescription approval, the exact sequence of approval checkpoints, the user interface requirements that enforce clinical oversight, and the audit points that document that oversight occurred.
This is not theoretical architecture — it is the operational specification for how AI-assisted peptide prescribing should function in a compliance-conscious, patient-safety-focused practice. Includes SOP templates, workflow diagrams, and required documentation standards.
I. The Core Workflow: Six Mandatory Checkpoints
AI-assisted peptide prescribing is not a linear process — it is a gated workflow where progression from one stage to the next requires explicit verification. Each gate serves a specific safety or compliance function. Bypass any gate, and the integrity of the entire process is compromised.
Workflow Diagram: AI-Assisted Peptide Prescribing with HITL Checkpoints
- PATIENT INTAKE
- DATA COMPLETENESS GATE
- AI RECOMMENDATION DRAFT
- CLINICIAN REVIEW SCREEN
- EXPLICIT APPROVAL CHECKPOINT
- ORDER ENTRY & MONITORING PLAN
We will now specify each checkpoint in operational detail.

Checkpoint 1: Patient Intake and Clinical Context Capture
Operational Requirement:
Before any AI recommendation can be generated, a complete patient clinical profile must be established. This is not merely data entry — it is the foundation of every subsequent decision.
Required Data Fields (Minimum):
- Patient demographics: Age, sex, weight (updated ≤30 days)
- Current medications: Complete list with start dates, reviewed and confirmed ≤7 days
- Known allergies: Drug and peptide-specific allergies documented
- Medical history: Renal function (eGFR), liver function (ALT/AST), cardiovascular history, pregnancy status (if applicable)
- Current peptide exposure: List of any peptides patient is currently taking, with lot numbers and start dates
- Treatment goals: What is the clinical indication for peptide therapy? (Document for ‘essentially a copy’ defense)
Gate Enforcement:
The system MUST prevent progression to AI recommendation generation if any mandatory field is missing or stale. Clinician should see a ‘Data Completeness Report’ showing missing or outdated fields, with action required before proceeding.
Checkpoint 2: Data Completeness Gate
Operational Requirement:
This is a quality control gate, not a clinical decision gate. The system verifies that the patient record contains all data required for safe AI-assisted prescribing.
Automatic Verification Checks:
- Freshness check: Are labs, vitals, and medication lists within acceptable age thresholds?
- Conflict detection: Are there any contradictions in the record (e.g., documented penicillin allergy but amoxicillin prescription)?
- Completeness validation: Are all mandatory fields populated?
- Unit consistency: Are all measurements in standard units (mg vs mcg, IU vs mg)?
Gate Enforcement:
- PASS: All checks satisfied → System allows progression to AI recommendation generation
- FAIL: One or more checks failed → System displays specific deficiencies and blocks progression until resolved
Checkpoint 3: AI Recommendation Draft Generation
Operational Requirement:
Only after Checkpoint 2 passes does the AI recommendation engine execute. This is where clinical intelligence is applied: peptide selection, dosing calculation, contraindication screening, monitoring protocol suggestion.
AI Output Components:
- Recommended peptide(s): Name, dose, frequency, duration
- Clinical rationale: Brief summary of why this peptide/dose is appropriate given patient context
- Evidence reference: Link to clinical guideline, research citation, or protocol source
- Risk flags surfaced: Any contraindications, cautions, or monitoring needs identified
- Alternative recommendations: Other viable peptide options considered and why they were ranked lower
Critical Design Principle:
The AI output is presented as a draft recommendation, not a prescription. The interface must not auto-populate prescription fields or create the impression that the recommendation is already approved. The physician must actively review and approve before any order is placed.
Related Resource: For comprehensive governance frameworks on AI clinical decision support: Human-in-the-Loop: AI Governance for Peptide Therapy
Checkpoint 4: Clinician Review Screen
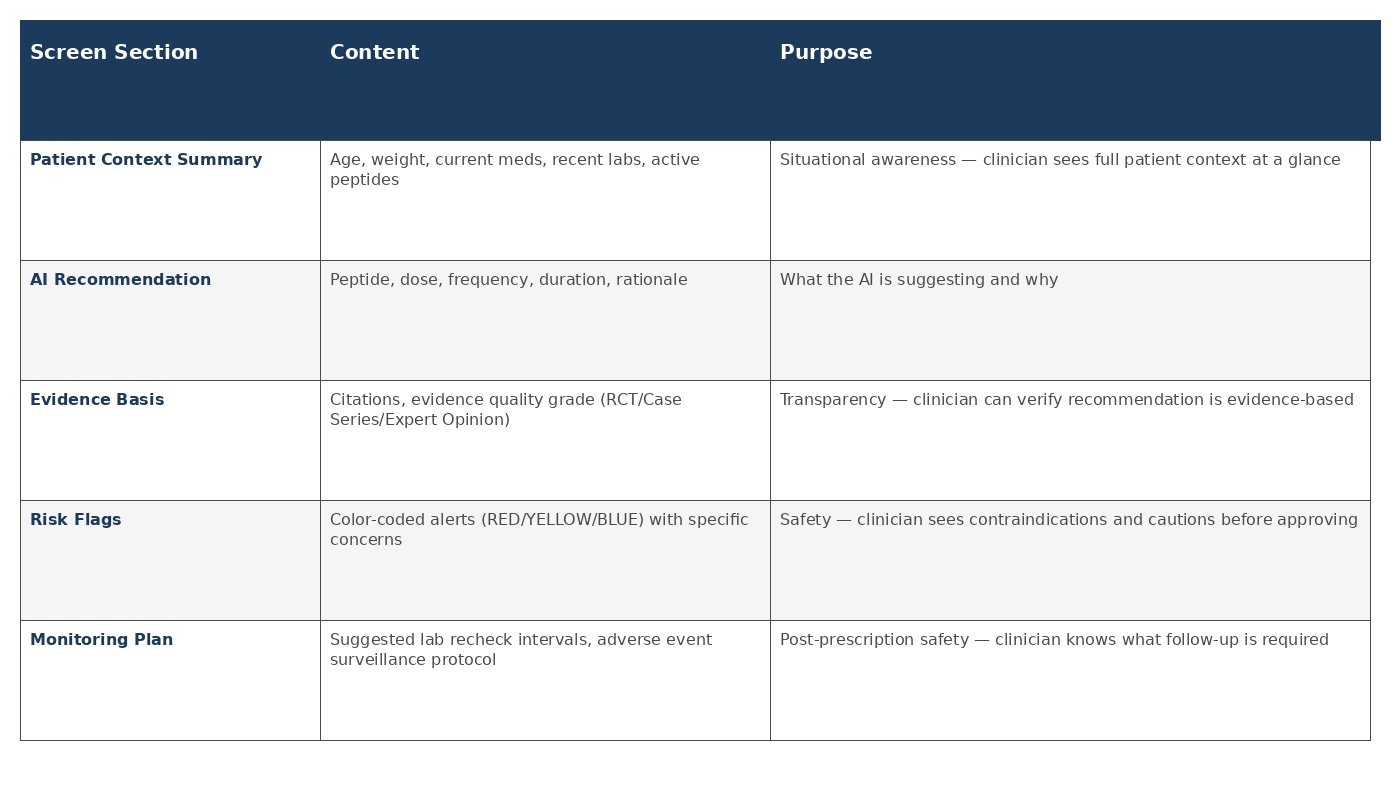
Operational Requirement:
This is the primary HITL checkpoint. The clinician is presented with a structured review interface that surfaces all clinically relevant information in a format optimized for rapid, informed decision-making.
Review Screen Components (Required UI Elements):
Interaction Design Principle:
The review screen CANNOT be bypassed. There is no ‘auto-approve’ option. The clinician must actively scroll through each section before the approval button becomes enabled. This is intentional friction — it forces engagement with the clinical reasoning.
Checkpoint 5: Explicit Approval Checkpoint
Operational Requirement:
This is the moment of clinical decision. The clinician must choose one of three actions, each with specific downstream consequences.

UI Design Requirements:
- Visual prominence: Approval action buttons must be large, clearly labeled, and visually distinct.
- Modification justification: If clinician selects ‘Approve With Modifications,’ a mandatory text field appears and must be completed before proceeding.
- Rejection justification: If clinician selects ‘Reject,’ a mandatory text field appears prompting for clinical reasoning.
- No default selection: None of the three action buttons should be pre-selected. The clinician must make an active choice.
✓ Prohibited UI Patterns (These Violate HITL Principles)
❌ One-click prescribing button that bypasses review
❌ Auto-populated prescription fields that require no clinician input
❌ ‘Approve All’ batch processing for multiple patients
❌ AI recommendation presented as ‘draft prescription’ rather than ‘draft recommendation’
❌ Bypass option that allows clinician to skip review screen ‘if familiar with protocol’
Checkpoint 6: Order Entry and Monitoring Plan Documentation
Operational Requirement:
Once approved, the system generates the prescription order and automatically creates a monitoring plan. Both must be documented in structured, auditable format.
Order Entry Components:
- Complete prescription: Patient name, peptide, strength, quantity, directions, prescriber signature, date
- Compounding pharmacy selection: Verified vendor from approved list (with current licensure confirmation)
- Lot number tracking: Space for lot number to be entered upon receipt (links prescription to vendor batch for outcome correlation)
- Informed consent documentation: Checkbox confirming patient was informed product is compounded and not FDA-approved
Monitoring Plan Components:
- Lab recheck schedule: Specific labs (e.g., ‘renal function panel’) with specific timing (e.g., ‘2 weeks, 1 month, 3 months’)
- Adverse event surveillance: Instructions for patient on what symptoms to report and how urgently
- Follow-up appointment: System schedules or prompts scheduling of follow-up visit to assess response and tolerance
The monitoring plan is not optional. It is generated automatically based on the peptide prescribed and patient risk factors, and is documented in the patient record for compliance verification.
Related Resource: For automated monitoring and safety surveillance infrastructure: Peptide Patient Safety: Automated Risk Flags
II. Standard Operating Procedure (SOP) Template
What follows is a template SOP that practices can adapt for their own use. It translates the six-checkpoint workflow into operational procedure language.
SOP: AI-Assisted Peptide Prescribing with Human-in-the-Loop Governance
Document Control:
SOP Number: [PRACTICE-ASSIGNED]
Effective Date: [DATE]
Review Frequency: Annually
Approvals Required: Medical Director, Compliance Officer
Supersedes: [PRIOR VERSION OR ‘N/A’]
1.0 Purpose
This SOP defines the standard workflow for AI-assisted peptide prescribing in [PRACTICE NAME]. It establishes mandatory checkpoints, approval requirements, documentation standards, and audit procedures to ensure clinical safety, regulatory compliance, and medicolegal defensibility.
2.0 Scope
This SOP applies to all prescribing clinicians using HolistiCare’s AI-assisted peptide protocol module. It covers peptide selection, dosing, contraindication screening, monitoring plan generation, and post-prescription surveillance.
3.0 Definitions
- AI Recommendation: Draft clinical suggestion generated by AI decision support system; not a prescription until clinician-approved
- HITL (Human-in-the-Loop): Governance model requiring explicit clinician approval before AI recommendations are executed
- Data Completeness Gate: Automated verification that patient record contains all required fields for safe prescribing
- Explicit Approval Checkpoint: Mandatory clinician action (Approve/Modify/Reject) with documented justification
4.0 Responsibilities
- Prescribing Clinician: Reviews AI recommendations, approves or modifies prescriptions, documents clinical reasoning, monitors patient outcomes
- Clinical Support Staff: Ensures patient intake data is complete and current before clinician review
- Medical Director: Conducts quarterly review of AI recommendation approval/rejection patterns, identifies protocol improvements
- Compliance Officer: Audits documentation completeness, monitors adverse events, maintains audit trail
5.0 Procedure
5.1 Pre-Prescribing: Patient Intake and Data Verification
- Clinical support staff completes patient intake, ensuring all mandatory data fields are current per Appendix A.
- Staff runs Data Completeness Check in HolistiCare system. If check fails, staff resolves deficiencies before escalating to prescribing clinician.
- Once Data Completeness Gate passes, case is flagged for prescribing clinician review.
5.2 AI Recommendation Generation
- Prescribing clinician opens patient record and initiates AI recommendation request for peptide therapy.
- System generates AI draft recommendation based on patient context, clinical guidelines, and contraindication database.
- AI recommendation includes: peptide selection, dosing parameters, clinical rationale, evidence citations, risk flags, monitoring plan.
5.3 Clinician Review and Approval
- Prescribing clinician reviews AI recommendation screen (cannot be bypassed).
- Clinician evaluates: patient context summary, AI recommendation details, evidence basis, risk flags, proposed monitoring plan.
- Clinician selects one of three actions:
- Approve As Recommended: Proceeds to order entry with AI-suggested parameters
- Approve With Modifications: Clinician edits parameters and documents modification rationale in mandatory text field
- Reject Recommendation: No prescription generated; clinician documents rejection rationale
5.4 Order Entry and Documentation
- If approved (with or without modifications), system generates prescription order with all required elements per Appendix B.
- Clinician selects approved compounding pharmacy vendor from verified list.
- System generates informed consent language confirming patient understands product is compounded and not FDA-approved. Clinician confirms consent discussion occurred.
- System automatically creates monitoring plan with lab recheck schedule and adverse event surveillance protocol. Clinician reviews and approves.
- Prescription is transmitted to pharmacy. Lot number field is flagged for completion upon product receipt.
5.5 Post-Prescribing: Monitoring and Surveillance
- System schedules automated reminders for lab rechecks per monitoring plan.
- Clinician documents patient response, tolerance, and any adverse events at each follow-up visit.
- If adverse event occurs, clinician follows Adverse Event Reporting SOP [CROSS-REFERENCE].
- If patient discontinues peptide, clinician documents reason (inefficacy, adverse effect, cost, patient preference, other).
6.0 Audit Points
The following data points are logged for every AI-assisted peptide prescription and are subject to quarterly audit:
- Timestamp: AI recommendation generated
- Timestamp: Clinician approval/modification/rejection
- AI recommendation details: Peptide, dose, rationale, evidence grade, risk flags
- Clinician decision: Approve/Modify/Reject
- Modification justification: If modified, clinician’s documented reasoning
- Rejection justification: If rejected, clinician’s documented reasoning
- Monitoring plan: Labs ordered, follow-up scheduled
- Outcome data: Patient response, adverse events, discontinuation reason
7.0 Quality Metrics
Medical Director reviews the following metrics quarterly:
- AI recommendation approval rate (target: 70-85%; if <70%, AI needs recalibration; if >95%, clinicians may not be exercising adequate independent judgment)
- Modification rate by peptide and by clinician (identifies protocol drift or training needs)
- Rejection rate and common rejection reasons (informs AI model improvement)
- Adverse event rate by peptide and by lot number (vendor quality surveillance)
- Monitoring plan adherence rate (% of patients who complete scheduled labs)
8.0 Revision History
[Table format: Version | Date | Author | Changes Made | Approvals]
9.0 Appendices
Appendix A: Mandatory Patient Data Fields and Freshness Requirements
Appendix B: Required Prescription Elements for Compounded Peptides
Appendix C: Approved Compounding Pharmacy Vendor List
Appendix D: Adverse Event Reporting Flowchart
Related Resource: For comprehensive peptide prescribing platform and workflow automation: HolistiCare Peptide Therapy Platform
III. Modification Logging and Audit Trail Requirements
Every deviation from AI recommendation must be logged with sufficient detail to support retrospective quality review, regulatory audit, and medicolegal defense.
3.1 What Must Be Logged
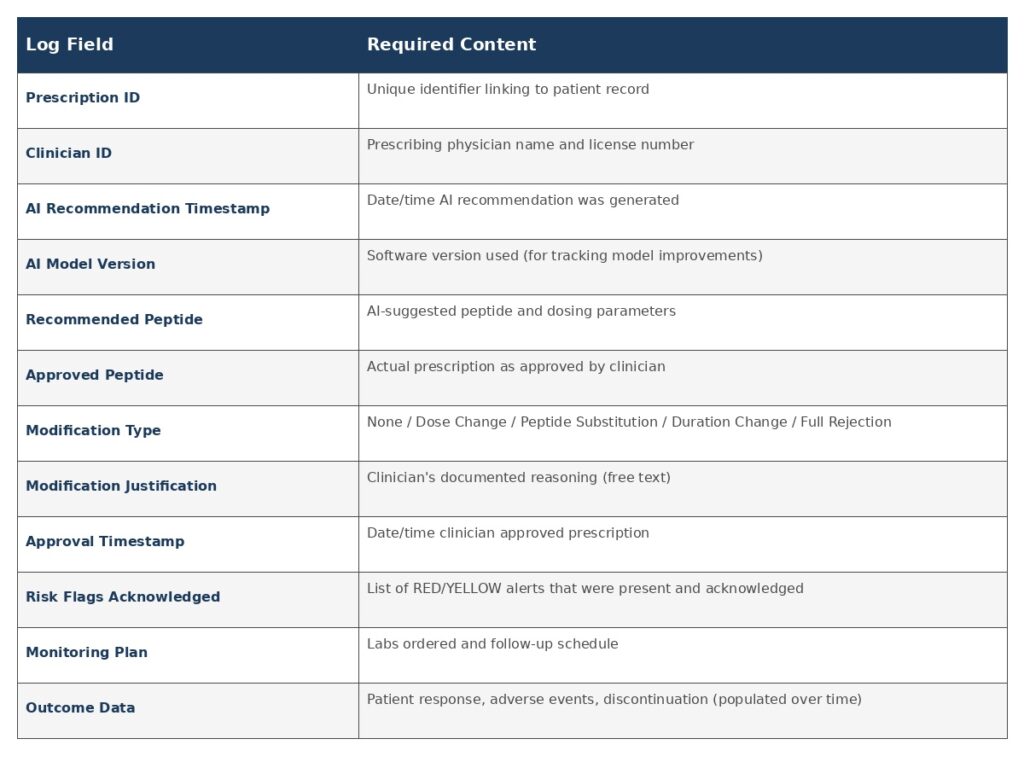
3.2 Audit Trail Immutability
Once a prescription decision is logged, the record CANNOT be edited or deleted. Corrections are appended as new entries with timestamp and attribution. This is critical for medicolegal defense — it demonstrates that records were not altered post-hoc in response to adverse events or regulatory inquiry.
3.3 Quarterly Audit Process
Medical Director or designee conducts structured audit each quarter:
- Pull all AI-assisted peptide prescriptions from prior quarter
- Review modification rate: Are clinicians modifying AI recommendations at expected rates? (Target: 15-30%)
- Sample 10 modified prescriptions: Read justifications. Are they clinically sound? Do they indicate protocol drift?
- Sample 5 rejected prescriptions: Why were they rejected? Is AI making inappropriate recommendations?
- Review adverse event correlation: Were any adverse events associated with prescriptions where AI flagged risks that clinician overrode?
- Document findings and corrective actions in Audit Report
- If patterns of concern emerge (e.g., high override rate on contraindication warnings), escalate to full clinical review and potential clinician retraining
IV. User Interface Requirements for HITL Enforcement
The workflow described above cannot be enforced through policy alone. It requires specific UI design patterns that make HITL checkpoints unavoidable.
4.1 Automated COA Validation and Anomaly Detection
The clinician review screen must implement the following UI enforcements:
Required UI Enforcements
- No bypass mechanism: Review screen cannot be closed or skipped without completing approval action.
- Scroll-to-enable: Approval buttons remain disabled until clinician has scrolled through all sections of review screen (Patient Context, AI Recommendation, Evidence, Risk Flags, Monitoring Plan).
- Timed minimum review: For high-risk prescriptions (flagged RED alerts), approval button remains disabled for minimum 30 seconds to prevent reflexive clicking.
- Visual risk hierarchy: RED/YELLOW/BLUE risk flags displayed with clear color coding and icons. RED flags require explicit acknowledgment checkbox before approval.
- Modification justification prompt: If clinician modifies any AI-recommended parameter, mandatory text field appears with prompt: ‘Document clinical reasoning for modification (required).
- Evidence access: Every AI recommendation includes ‘View Evidence’ link that opens supporting citations in expandable panel. Clinician can review without navigating away from approval screen.
4.2 Modification Logging Interface
When clinician selects ‘Approve With Modifications,’ the system must present:
- Side-by-side comparison: AI Recommended vs Clinician Modified (e.g., ‘AI Rec: BPC-157 500mcg BID’ | ‘Modified: BPC-157 250mcg BID’)
- Mandatory justification field: Free text, minimum 10 characters, prompt: ‘Why are you modifying this recommendation?’
- Common modification reasons (checkboxes + other): Patient tolerance concern / Prior response data / Drug interaction / Clinician experience / Other (specify)
- Explicit acknowledgment: ‘I have reviewed the AI recommendation and evidence basis, and I am exercising independent clinical judgment to modify this prescription.’
4.3 Mobile and Tablet Considerations
Many clinicians review cases on mobile devices. The HITL workflow must function correctly on small screens without compromising safety checkpoints:
- Responsive design: Review screen sections collapse into accordion panels on mobile, but all panels must still be expanded and reviewed before approval
- Voice-to-text for justifications: Mobile justification fields should support voice input to reduce typing burden
- Offline capability: If connectivity is lost mid-review, system caches patient data and AI recommendation, allows clinician to complete review, and syncs when connectivity restored (with timestamp reflecting actual approval time)
- No approval via mobile notification: Clinicians should not receive push notifications with ‘Approve this prescription?’ buttons. Approval must occur within the full review interface.
V. Training Requirements and Clinician Onboarding
The workflow is only as effective as the clinicians’ understanding of it. Comprehensive training is mandatory before clinicians are granted access to AI-assisted prescribing tools.
5.1 Pre-Deployment Training Modules (Required)
- Module 1: HITL Governance Principles (30 min): Why AI cannot autonomously prescribe; what can go wrong when humans defer to algorithms without review; case studies of AI recommendation errors caught by clinician review
- Module 2: Workflow Walkthrough (45 min): Step-by-step demonstration of six-checkpoint workflow; hands-on practice with test cases
- Module 3: Risk Flag Interpretation (30 min): What RED/YELLOW/BLUE flags mean; how to evaluate whether a flagged risk is clinically significant for this specific patient
- Module 4: Modification Documentation (20 min): How to write clinically sound and legally defensible modification justifications; examples of good vs. poor documentation
- Module 5: Audit Trail and Compliance (15 min): What gets logged, why it matters, what regulators/litigators will look for in records
5.2 Competency Assessment
All clinicians using AI-assisted prescribing must complete annual refresher training covering:
- Updates to AI model capabilities and limitations
- New peptides added to formulary or bulk substance list
- Recent FDA enforcement actions or regulatory guidance affecting compounding
- Review of practice’s own audit data: common modification patterns, adverse event correlation, areas for improvement
5.3 Annual Refresher Training
Before clinicians are approved to use AI-assisted prescribing in live patient care, they must complete a competency assessment:
- 5 simulated patient cases with AI recommendations
- Clinician must review each case, identify risk flags, and decide whether to approve, modify, or reject
- Clinician must document justification for any modifications or rejections
- Passing score: 4/5 cases with clinically appropriate decisions and adequate documentation
- If clinician fails, remediation training required before retesting
VI. Conclusion: Workflow as the Enforcement Layer of Governance
The HITL governance framework is not merely a set of principles — it is an operational specification that must be encoded into clinical workflow. The six-checkpoint workflow described in this guide is the enforcement mechanism. Without it, HITL governance is aspirational. With it, HITL governance is structural.
The SOP template, UI requirements, modification logging standards, and training modules are not optional enhancements. They are the minimum operational infrastructure required to deploy AI-assisted peptide prescribing in a manner that is clinically responsible, legally defensible, and institutionally sustainable.
Practices that implement this workflow will discover that it does not slow down prescribing — it accelerates it by surfacing the right information at the right time and documenting compliance automatically. Practices that attempt to deploy AI clinical decision support without this workflow will discover that they have created liability faster than they have created value.
HolistiCare’s platform implements every element of this workflow as designed — from data completeness gates through forced approval screens through immutable audit logging. This is not marketing. This is operational reality, refined through thousands of prescriptions across our partner practices.
Build the workflow correctly. Enforce the checkpoints rigorously. Document everything. The alternative is to operate AI-assisted prescribing on trust and vigilance rather than on architecture and enforcement — and that is not a position any serious practice should accept.
References
[1] Topol, E. J. (2019). High-performance medicine: the convergence of human and artificial intelligence. Nature Medicine, 25(1), 44-56.
[2] Char, D. S., Shah, N. H., & Magnus, D. (2018). Implementing Machine Learning in Health Care — Addressing Ethical Challenges. New England Journal of Medicine, 378(11), 981-983.
[3] FDA. (2021). Artificial Intelligence and Machine Learning in Software as a Medical Device. FDA Guidance for Industry.
[4] Obermeyer, Z., Powers, B., Vogeli, C., & Mullainathan, S. (2019). Dissecting racial bias in an algorithm used to manage the health of populations. Science, 366(6464), 447-453.
[5] Rajkomar, A., Dean, J., & Kohane, I. (2019). Machine Learning in Medicine. New England Journal of Medicine, 380(14), 1347-1358.
[6] Institute of Medicine. (2000). To Err is Human: Building a Safer Health System. National Academies Press.
[7] Bates, D. W., Kuperman, G. J., Wang, S., et al. (2003). Ten Commandments for Effective Clinical Decision Support. Journal of the American Medical Informatics Association, 10(6), 523-530.
[8] Sittig, D. F., & Singh, H. (2010). A new sociotechnical model for studying health information technology in complex adaptive healthcare systems. Quality and Safety in Health Care, 19(Suppl 3), i68-i74.
Legal & Medical Disclaimer:
This document is produced for educational and operational guidance purposes by HolistiCare.io and does not constitute legal advice, regulatory counsel, medical practice standards, or clinical protocols. The workflow specifications, SOP templates, UI requirements, and training modules presented are operational frameworks intended for adaptation by qualified healthcare professionals in consultation with legal counsel, compliance officers, and clinical leadership. Implementation of the described workflow does not guarantee regulatory compliance, clinical safety, or legal protection from liability. All clinical decision-making remains the sole responsibility of licensed prescribing physicians. AI-assisted clinical decision support systems must be deployed in accordance with applicable FDA regulations, state medical board requirements, institutional policies, and professional standards of care. Practices are advised to consult qualified legal counsel, regulatory compliance specialists, and clinical risk management professionals before implementing AI-assisted prescribing workflows or modifying existing clinical protocols. The SOP template provided is a starting framework and must be customized to reflect practice-specific operations, state-specific regulations, and institutional policies. HolistiCare.io makes no representations regarding the adequacy, completeness, or legal sufficiency of the provided templates for any specific practice or jurisdiction. HolistiCare.io is a clinical intelligence software company and does not provide direct clinical services, legal advice, regulatory consulting, or clinical practice management services.






