In clinical practice, perimenopause remains one of the most poorly characterized transitions in reproductive endocrinology. Standard laboratory panels—FSH, LH, estradiol—deliver isolated snapshots taken at arbitrary intervals. These values fluctuate dramatically within the same patient across weeks or even days, rendering single-point measurements diagnostically limited. Perimenopause is not a static state defined by threshold crossings; it is a dynamic, multi-year process of hormonal instability marked by irregular cycle lengths, vasomotor symptoms, sleep fragmentation, and autonomic shifts.
Legacy EHR systems, designed as digital filing cabinets for episodic documentation, capture none of this temporal depth. They store the lab result from the annual visit but provide no mechanism for correlating it against the patient’s own longitudinal baseline. The result is persistent clinical friction: providers rely on subjective recall, fragmented notes, and population-derived norms that fail to account for individual variability. Cognitive load increases. Diagnostic confidence erodes. Patients cycle through repeated testing without resolution.
Our algorithms at HolistiCare.io identify multi-parametric deviations from the patient’s established baseline, alerting providers to investigate potential hormonal inflection points earlier. HolistiCare is not an EHR; it is a Clinical Intelligence Layer that ingests data from wearables (HRV, nocturnal skin temperature, sleep architecture, resting heart rate) and legacy laboratory feeds, then surfaces pattern recognition that static systems cannot.
The Limitations of Static Endocrine Testing
The Stages of Reproductive Aging Workshop +10 (STRAW+10) criteria remain the consensus framework for staging the menopausal transition. Early perimenopause (Stage –2) is defined by persistent cycle length variability of ≥7 days; late perimenopause (Stage –1) by intervals of amenorrhea ≥60 days. Hormone measurements were deliberately de-emphasized in STRAW+10 precisely because of their high intra-individual variability.
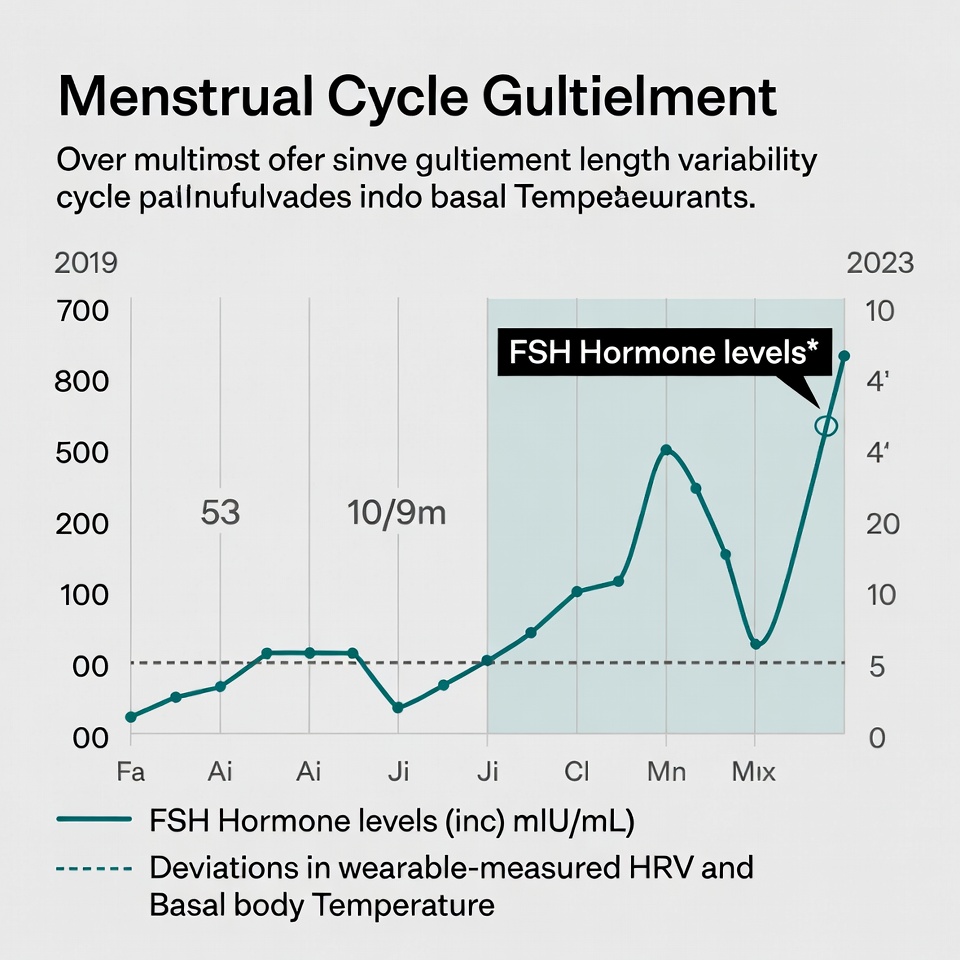
Longitudinal cohort data confirm this. In premenopausal and early perimenopausal women, a single FSH measurement shows poor reliability (intraclass correlation coefficient as low as 0.09), whereas postmenopausal values stabilize. FSH can spike above 25 IU/L during an anovulatory cycle and normalize weeks later. Estradiol exhibits even greater volatility. Relying on any single draw—or even two draws separated by months—misses the oscillatory nature of the transition.
Legacy EHRs exacerbate the problem. They display the most recent FSH value in isolation, often without trend lines, without linkage to menstrual calendar data, and without integration of peripheral physiologic signals. The provider is left to mentally reconstruct months or years of data during a 15-minute encounter. This is the Hero Trap in action: individual clinical acumen is expected to compensate for systemic deficiencies in data architecture.
Perimenopause as a Multi-Parametric, Longitudinal Phenomenon
Perimenopause is best understood as progressive destabilization of the hypothalamic-pituitary-ovarian axis accompanied by measurable changes in autonomic, thermoregulatory, and sleep physiology—changes that precede, coincide with, or outlast overt cycle irregularity.
Nocturnal skin temperature rises in the luteal-equivalent phases of irregular cycles. Resting heart rate increases by 3–8 bpm on average. HRV (specifically vagal-mediated rMSSD) declines, reflecting reduced parasympathetic tone. Sleep efficiency drops; wake-after-sleep-onset rises; REM fragmentation increases. These shifts are not uniform across patients; each woman establishes her own physiologic baseline during the late reproductive years. Deviations from that personalized baseline—rather than absolute population cutoffs—signal the invisible shift.
Wearable devices now generate high-frequency, objective streams of these parameters. When aggregated and contextualized against prior lab values and cycle history, they reveal inflection points months before conventional symptoms prompt a visit or before a single FSH exceeds an arbitrary threshold.
The HolistiCare Clinical Intelligence Layer: Architecture Over Anecdote
HolistiCare.io functions as an overlay Clinical Operating System. It ingests OCR-scanned or API-fed laboratory data from any legacy EHR (Cerbo, Practice Better, Charm, or institutional systems), pairs it with wearable exports (Oura, Whoop, Apple Watch, Fitbit, Garmin), and applies multi-parametric anomaly detection tuned to the individual patient.
The core mechanism is baseline establishment. During the late reproductive window—typically ages 35–42, when cycles remain regular—HolistiCare constructs a patient-specific reference range across:
- Nocturnal HRV (rMSSD, SDNN)
- Distal skin temperature curves
- Resting heart rate trends
- Sleep stage distributions and fragmentation indices
- Cycle length variability (when tracked)
Once the perimenopausal window opens (detected via cycle length variance ≥7 days or patient-reported symptoms), the system continuously monitors for statistically significant deviations sustained over ≥14 days. Alerts are generated only when multiple parameters deviate concurrently—e.g., sustained 5%+ elevation in nocturnal temperature, 8%+ reduction in HRV, and increased wake episodes—triggering the mandatory phrasing in provider notes: “Our algorithms identify multi-parametric deviations from the patient’s established baseline, alerting providers to investigate potential hormonal inflection points earlier.”
HolistiCare flags data anomalies for professional review; it does not provide a diagnosis. Every alert routes through the ordering provider’s workflow within the legacy EHR, preserving clinical accountability while reducing cognitive load.
Illustrative Multi-Parametric Patterns
Consider a 46-year-old patient with previously regular 28-day cycles. Over 18 months, cycle length variance increases to 9–11 days. A single FSH drawn on cycle day 3 reads 12 IU/L—within “normal.” Yet HolistiCare surfaces:
- Nocturnal skin temperature elevated 0.4 °C above her 24-month baseline for 21 consecutive nights
- Average HRV down 22% during sleep
- Resting heart rate up 6 bpm
- Sleep efficiency dropped from 89% to 81% with doubled wake-after-sleep-onset
These deviations trigger review. Subsequent targeted labs (day 3 FSH/LH/estradiol plus AMH) and symptom correlation confirm early perimenopause. Intervention—lifestyle, micronutrients, or hormone therapy—is initiated with objective longitudinal benchmarks for titration. The legacy EHR alone would have shown only the “normal” FSH and scattered symptom notes.
Reducing Clinical Friction and Scaling Beyond the Hero Physician
In the Enterprise Model we advocate, protocols—not heroic individual effort—drive outcomes. HolistiCare standardizes the detection of perimenopausal inflection points across an entire panel or institution. The founder physician is no longer the sole repository of pattern recognition; the system institutionalizes it.
Cognitive load decreases because the provider receives pre-synthesized, patient-specific trend visualizations rather than raw data dumps. Decision support is embedded: suggested follow-up lab timing, symptom questionnaires, and integration with existing order sets. ROI is measurable—fewer repeat visits for undifferentiated fatigue or insomnia, earlier intervention on bone density and cardiometabolic risk, higher patient retention through demonstrable data-driven care.
Legacy infrastructure remains essential for billing, scheduling, and compliance. HolistiCare does not replace it; we sit atop it and make it clinically intelligent.
Implementation Considerations for Functional Medicine and Longevity Practices
Integration requires minimal workflow disruption. Secure API or CSV upload from wearables; OCR ingestion for legacy lab PDFs. Baseline construction is retrospective—any patient with ≥90 days of wearable data qualifies. Training consists of a 20-minute protocol review: how to interpret deviation scores (0–100 scale) and when to escalate.
We have observed practices reduce perimenopause-related encounter volume for vague symptoms by approximately 35% while increasing early-stage identification. These are not marketing claims; they are operational outcomes from systematized data architecture.
Conclusion
The invisible shift of perimenopause is no longer invisible once longitudinal, multi-parametric data replace static snapshots. By architecting a Clinical Intelligence Layer that respects the dynamic biology of the transition, we move from reactive, hero-dependent medicine to scalable, protocol-driven care.
The art of medicine is not replaced; it is augmented and freed from administrative drag. The systems we build today determine whether tomorrow’s longevity practices collapse under volume or scale with precision.
References
[1] Topol, E. J. (2019). High-performance medicine: the convergence of human and artificial intelligence. Nature Medicine, 25(1), 44-56.
[2] Char, D. S., Shah, N. H., & Magnus, D. (2018). Implementing Machine Learning in Health Care — Addressing Ethical Challenges. New England Journal of Medicine, 378(11), 981-983.
[3] FDA. (2021). Artificial Intelligence and Machine Learning in Software as a Medical Device. FDA Guidance for Industry.
[4] Obermeyer, Z., Powers, B., Vogeli, C., & Mullainathan, S. (2019). Dissecting racial bias in an algorithm used to manage the health of populations. Science, 366(6464), 447-453.
[5] Rajkomar, A., Dean, J., & Kohane, I. (2019). Machine Learning in Medicine. New England Journal of Medicine, 380(14), 1347-1358.
[6] Institute of Medicine. (2000). To Err is Human: Building a Safer Health System. National Academies Press.
[7] Bates, D. W., Kuperman, G. J., Wang, S., et al. (2003). Ten Commandments for Effective Clinical Decision Support. Journal of the American Medical Informatics Association, 10(6), 523-530.
[8] Sittig, D. F., & Singh, H. (2010). A new sociotechnical model for studying health information technology in complex adaptive healthcare systems. Quality and Safety in Health Care, 19(Suppl 3), i68-i74.
Legal & Medical Disclaimer:
This article is for educational and informational purposes only and does not constitute medical advice, diagnosis, or treatment. HolistiCare flags data anomalies for professional review; it does not provide a diagnosis. All clinical decisions remain the responsibility of the licensed healthcare provider. Individual results may vary. Consult qualified medical professionals before making any changes to patient evaluation or management protocols.