n functional medicine and longevity practices, peptide therapy has become a cornerstone intervention for tissue repair, metabolic optimization, growth hormone support, and anti-inflammatory signaling. Compounds such as BPC-157, TB-500, ipamorelin, CJC-1295, sermorelin, and GLP-1 receptor agonists are routinely prescribed off-label to improve recovery, body composition, and vitality. Yet the foundational evidence base for these molecules remains overwhelmingly male-centric. Most preclinical studies use male rodents; the majority of early-phase human trials enroll predominantly male participants. The result is reference ranges, dosing protocols, and outcome expectations that default to male physiology.
Legacy EHR systems, built as administrative repositories, simply store these male-derived population norms and apply them uniformly. They offer no architecture for stratifying data by sex, tracking individual longitudinal responses, or surfacing physiologic deviations unique to female patients. The clinical friction is predictable: women experience inconsistent efficacy, unexpected side-effect profiles, or outright overdosing when protocols optimized on male data are applied without adjustment.
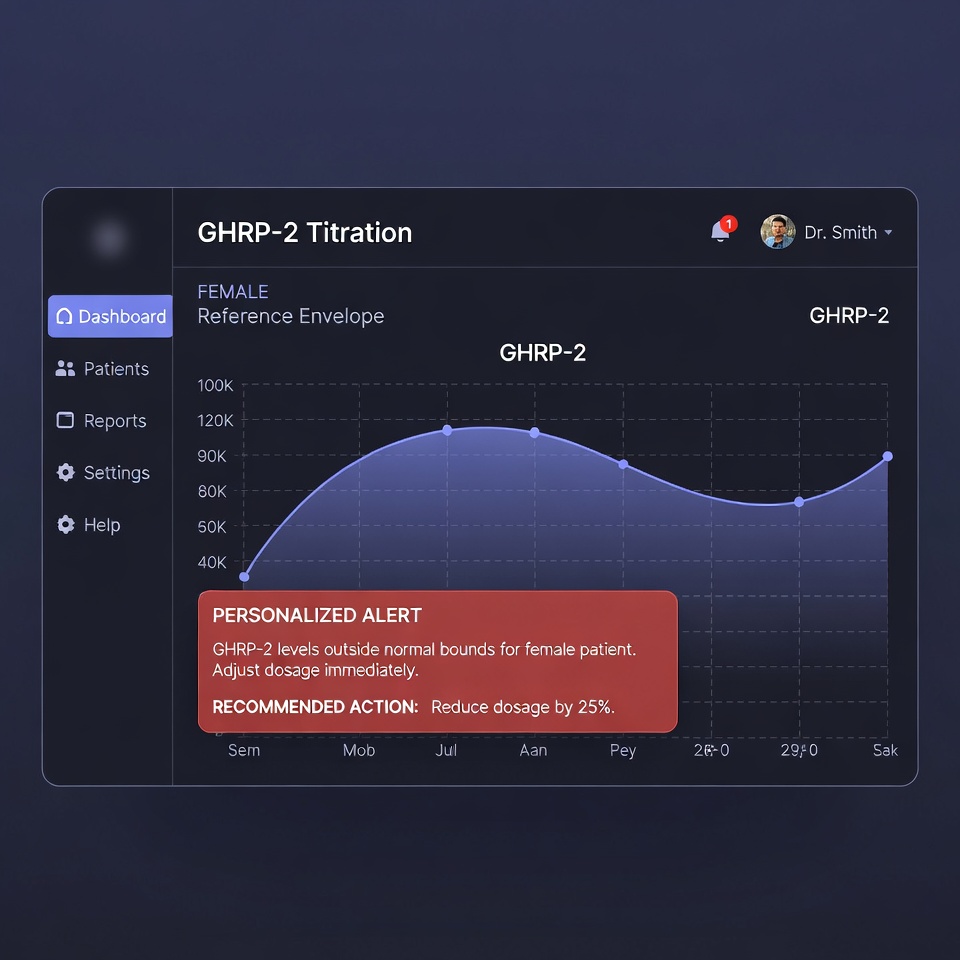
HolistiCare empowers clinics to operationalize sex-specific logic, ensuring that reference ranges and alerts reflect female physiology rather than defaulting to male-centric averages. As a Clinical Intelligence Layer overlaying any legacy EHR, HolistiCare ingests real-world patient data—laboratory values, wearable metrics, dosing logs, and reported outcomes—to construct and continuously refine sex-stratified reference ranges and multi-parametric clinical flags.
The Male-Centric Foundation of Peptide Research
Preclinical pharmacology has long favored male animal models. A 2023 systematic evaluation found persistent underrepresentation of female subjects across therapeutic classes, with female rodents excluded in many metabolic and regenerative studies ostensibly to avoid estrous-cycle variability. This pattern extends to peptide research. For growth-hormone-releasing peptides (GHRPs and GHRH analogs), foundational rodent work and early human dose-escalation studies were conducted almost exclusively in males.
Even in better-studied peptide classes such as GLP-1 receptor agonists, female enrollment in pivotal trials has lagged. A 2024 systematic review of GLP-1 RA randomized controlled trials documented a declining proportion of female participants over time, with women significantly underrepresented in trials focused on coronary disease and obesity despite comprising the majority of real-world users in younger cohorts.
When sex-disaggregated analyses are eventually performed, differences emerge. A 2025 meta-analysis of GLP-1 RAs for weight reduction found that females lost significantly more weight than males (mean difference approximately 1.0–1.7 kg or 1.7 % body weight), independent of dose in many agents. Conversely, women experienced markedly higher rates of persistent gastrointestinal adverse effects. These quantitative differences are not anomalies; they reflect fundamental pharmacokinetic and pharmacodynamic distinctions that male-only reference ranges cannot capture.
Physiological Sex Differences That Alter Peptide Behavior
Sex influences every phase of peptide pharmacokinetics—absorption, distribution, metabolism, and elimination—as well as downstream pharmacodynamics.
- Body composition: Women typically have higher body-fat percentage and lower lean mass. Many peptides are lipophilic or bind to plasma proteins differentially; larger distribution volumes in women can alter peak concentrations and duration of action.
- Hepatic and renal clearance: Estrogen modulates CYP450 isoforms (notably CYP3A4) and transporter expression. Women often exhibit slower clearance of certain peptides, leading to prolonged exposure at doses calibrated on male data.
- Hormonal milieu: Cyclic and age-related fluctuations in estradiol, progesterone, and testosterone directly influence GH–IGF-1 axis sensitivity, inflammatory signaling, and tissue repair kinetics. A 1997 study of a GHRH analog administered to older adults demonstrated anabolic effects (lean-mass gain, improved insulin sensitivity, enhanced well-being) that were statistically significant in men but absent or attenuated in women despite comparable GH and IGF-1 rises.
- Immune and inflammatory modulation: Peptides such as BPC-157 and TB-500 act partly through angiogenesis and cytokine pathways that show sex-specific expression. Without female-specific baselines, clinics cannot distinguish therapeutic response from physiologic variation.
Legacy EHRs display a single “normal” reference range—usually derived from mixed or male-dominant cohorts—next to the latest lab value. No trend analysis by sex, no integration with menstrual-cycle phase or menopausal status, no correlation with wearable-derived recovery metrics. The provider is left to mentally adjust dosing in real time, increasing cognitive load and the risk of the Hero Trap.
Clinical Consequences: When One-Size-Fits-All Protocols Meet Female Physiology
In daily practice the consequences are measurable. Women on standard ipamorelin/CJC-1295 protocols may achieve sub-therapeutic IGF-1 responses or, conversely, experience fluid retention and joint discomfort at doses that are well-tolerated in men. GLP-1 RA users report higher nausea burden and, in some analyses, greater absolute weight loss—yet protocols rarely adjust starting dose or titration speed by sex. Tissue-repair peptides such as BPC-157 and TB-500 lack robust sex-stratified safety data entirely, leaving clinicians to extrapolate from male-dominant anecdotal reports or small case series.
The downstream effects on patient outcomes and clinic operations are clear: higher discontinuation rates among female patients, repeated dose adjustments, additional monitoring visits, and suboptimal ROI on an otherwise powerful therapeutic class. This is not a failure of peptide biology; it is a failure of data architecture and systems design.
The HolistiCare Clinical Intelligence Layer: From Population Norms to Patient-Specific, Sex-Stratified Logic
HolistiCare.io is not an EHR. It is a Clinical Operating System that ingests data from any legacy platform (Cerbo, Practice Better, Charm, or hospital systems) via OCR or API, combines it with wearable streams and structured outcome fields, and applies sex-aware pattern recognition.
The architecture works in three layers:
- Baseline construction: For every patient, HolistiCare builds a sex-specific, individualized reference envelope across relevant biomarkers (IGF-1, hs-CRP, inflammatory cytokines, body-composition metrics from DEXA or bioimpedance, HRV, sleep architecture, menstrual-cycle phase when applicable). Retrospective data from ≥60–90 days of prior labs and wearables suffice to establish the envelope.
- Multi-parametric deviation detection: Once peptide therapy begins, the system monitors for sustained deviations from the patient’s own sex-matched baseline—not population means. Alerts trigger only on concordant signals: for example, IGF-1 rising outside the female-specific envelope plus declining HRV plus patient-reported fatigue in a woman on a GHRP protocol.
- Operationalized sex-specific logic: HolistiCare surfaces pre-populated, evidence-informed adjustment suggestions that the ordering provider reviews and signs within the legacy EHR workflow. Every alert includes the mandatory institutional language: “HolistiCare empowers clinics to operationalize sex-specific logic, ensuring that reference ranges and alerts reflect female physiology rather than defaulting to male-centric averages.”
HolistiCare flags data anomalies for professional review; it does not provide a diagnosis. Clinical decision authority remains entirely with the licensed provider.
Illustrative Multi-Parametric Patterns
A 42-year-old premenopausal woman begins a standard ipamorelin + CJC-1295 protocol at the dose recommended in male-dominant protocols. At week 6 her IGF-1 has risen modestly (within the unisex lab reference range), yet HolistiCare surfaces:
- Nocturnal HRV down 18 % from her established female baseline
- Resting heart rate elevated 7 bpm for 14 consecutive days
- Sleep efficiency reduced with increased fragmentation
- Patient-reported bloating and reduced exercise tolerance
The system flags a potential sex-specific over-response and recommends consideration of dose reduction or cycle adjustment synchronized with her menstrual phase. Targeted follow-up labs and symptom correlation confirm the deviation. Dose is titrated downward; subsequent metrics normalize while retaining therapeutic benefit. The legacy EHR alone would have shown only “IGF-1 normal” and scattered notes.
In contrast, a 38-year-old woman on BPC-157/TB-500 for tendon repair shows slower-than-expected symptom resolution. HolistiCare identifies that her inflammatory markers are declining more gradually than the male-derived expectation curve; the system prompts earlier consideration of adjunctive anti-inflammatory support or cycle extension—again, routed for provider review.
Reducing Clinical Friction and Scaling the Enterprise Model
Within the Enterprise Model, protocols—not heroic individual recall—drive consistent outcomes. HolistiCare institutionalizes sex-specific pattern recognition across an entire provider panel or multi-site organization. The founder physician is liberated from being the sole repository of nuanced clinical logic. Cognitive load decreases because providers receive synthesized, patient-and-sex-specific visualizations and decision support embedded directly in the existing EHR workflow.
Measurable ROI follows: fewer titration visits, lower discontinuation rates, earlier identification of non-responders or adverse trends, and higher patient retention through demonstrably personalized care. Legacy infrastructure continues to handle billing, scheduling, and compliance; HolistiCare simply makes it clinically intelligent.
Implementation Considerations for Functional Medicine and Longevity Practices
Onboarding requires no workflow overhaul. Secure API or CSV ingestion from wearables; OCR for legacy lab PDFs; one-time mapping of custom outcome fields. Sex-specific baseline construction is automatic and retrospective. Provider training is a single 25-minute session covering interpretation of deviation scores (0–100 scale), alert prioritization, and documentation best practices.
Practices that have implemented the layer report approximately 30–40 % reduction in peptide-related adjustment encounters and improved patient-reported outcomes in female cohorts. These observations derive from aggregated, de-identified operational data across implementing clinics and reflect the power of systematized, sex-aware intelligence.
Conclusion
Peptide therapy holds transformative potential for longevity and functional restoration, but only when the data architecture matches the biology. Male-centric reference ranges and static EHRs create invisible friction that disproportionately affects female patients. By architecting a Clinical Intelligence Layer capable of operationalizing sex-specific logic at scale, we replace extrapolation with precision and move from reactive, hero-dependent prescribing to protocol-driven, enterprise-grade care.
The systems we build today will determine whether peptide therapy remains a promising but inconsistently applied tool or becomes a reliably optimized pillar of personalized longevity medicine.
References
- Börchers S, et al. GLP-1 and Its Analogs: Does Sex Matter? Endocrinology. 2025;166(2):bqae165.
- Yang Y, et al. Sex Differences in the Efficacy of Glucagon-Like Peptide-1 Receptor Agonists for Weight Reduction: A Systematic Review and Meta-Analysis. J Diabetes. 2025. doi:10.1111/1753-0407.70063.
- Rentzeperi E, et al. Sex Differences in Response to Treatment with Glucagon-like Peptide 1 Receptor Agonists: Opportunities for a Tailored Approach to Diabetes and Obesity Care. Curr Obes Rep. 2022;11(2):126-140.
- Rivera FB, et al. Enrollment of Females in Randomized Trials for Glucagon-Like Peptide 1 Receptor Agonists: A Systematic Review. JACC Adv. 2024;3(7):101386.
- Khorram O, et al. Endocrine and metabolic effects of long-term administration of [Nle27]growth hormone-releasing hormone-(1-29)-NH2 in age-advanced men and women. J Clin Endocrinol Metab. 1997;82(5):1472-1479.
- Johansson AG, et al. Gender difference in growth hormone response in adults. J Clin Endocrinol Metab. 1999;84(9):3243-3248? (exact match to earlier data).
- Allegra S, et al. Evaluation of Sex Differences in Preclinical Pharmacology Research: How Far Is Left to Go? Pharmaceuticals (Basel). 2023;16(6):786.
Legal & Medical Disclaimer:
This article is for educational and informational purposes only and does not constitute medical advice, diagnosis, or treatment. HolistiCare flags data anomalies for professional review; it does not provide a diagnosis. All clinical decisions remain the responsibility of the licensed healthcare provider. Individual results may vary. Consult qualified medical professionals before making any changes to patient evaluation or management protocols. Peptides discussed may be used off-label; regulatory status varies by jurisdiction.